View Procedure
| Procedure Name | Genetically Modified Organism (GMO) Import/Export/Transit Permit | |||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Description |
Required Documents
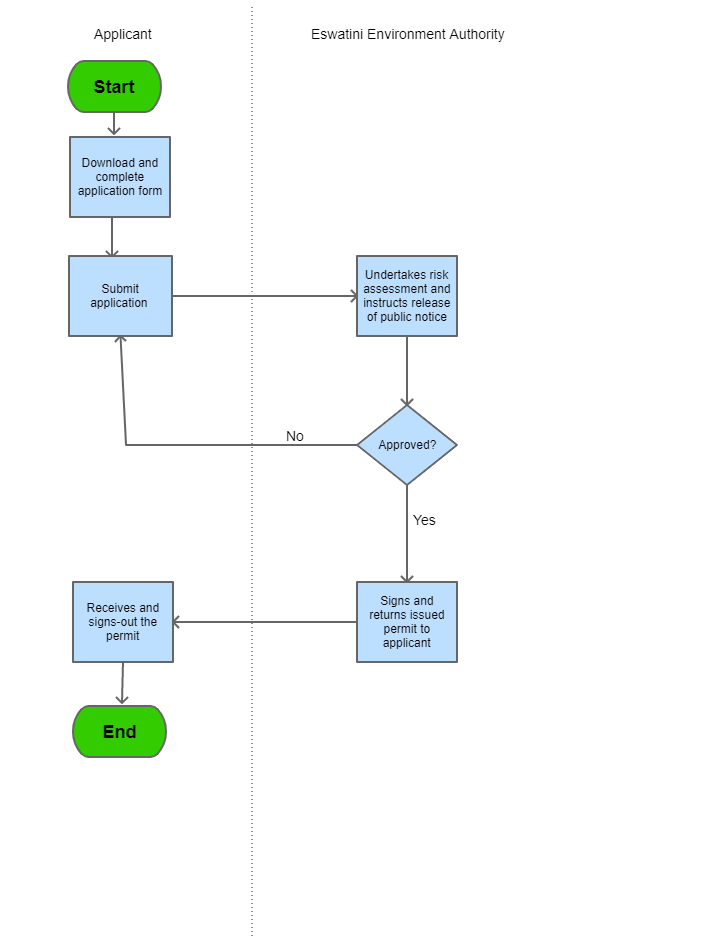
Process Steps
| |||||||||||||||||||||||||||||||||||||||
| Category | Import |
The following form/s are used in this procedure
| Title | Description | Created Date | Updated Date | Issued By |  |
|---|---|---|---|---|---|
| Application Form For Import, Export And Transit Of Genetically Modified Organisms | Application Form For Import, Export And Transit Of Genetically Modified Organisms | 11-06-2020 | 11-06-2020 |
This procedure applies to the following measures
| Name | Measure Type | Agency | Description | Comments | Legal Document | Validity To | Measure Class |
|---|---|---|---|---|---|---|---|
| GMO Import Permit | Permit Requirement | This process represents the procedure for obtaining a permit to import genetically modified Organisms (GMO), most typically crops represented by the import of cotton seed, maize seed and maize grain in to the Kingdom of Eswatini. All GMOs are controlled for the protection of the environment and human health. | Depending on the product, the issued permits have a variable duration. | The Biosafety Act, 2012 | 31-12-2030 | Good | |
| Documentation Requirements for Genetically Modified Organism | Permit Requirement | Documentation for GMOs intended for contained use 1. GMOs that are imported into or exported from the country for contained use shall be accompanied by documentation that- (a) clearly identifies the GMOs as GMOs; (b) specifies any requirements for the safe handling, storage, transport and use; and (c) provides a contact point for further information, including the name and address of the individual and institution to whom the living modified organisms are consigned. 2. Any additional documentation or identification requirements applicable to imports or exports subject to subsection (1) and agreed upon under the Cartagena Protocol shall be addressed by regulation in accordance with section 34. Documentation for GMOs for direct use as food, feed or for processing 1. Documentation that clearly identifies that the goods “may contain” GMOs and are not intended for intentional introduction into the environment. 2. Contact point for further information. 3. Any additional documentation or identification requirements applicable to imports or exports subject to subsection (1) and agreed upon under the Cartagena Protocol shall be addressed by regulation in accordance with section 34. Documentation for GMOs intended for intentional introduction into the environment 1. GMOs that are imported into or exported from the country for intentional introduction into the environment shall be accompanied by documentation that- a. clearly identifies the GMOs as GMOs; b. specifies the identity and relevant traits and characteristics, any requirements for the safe handling, storage, transport and use, the contact point for further information and, as appropriate, the name and address of the importer or exporter; c. contains a declaration that the movement is in conformity with the requirements of the Cartagena Protocol applicable to the exporter. 2. Any additional documentation or identification requirements applicable to imports or exports subject to subsection (1) (a) and agreed upon under the Cartagena Protocol shall be addressed by regulation in accordance with section 34. | Requirements necessary for the documentation and labeling of GMOs intended for; a. import and export, b. direct use as food, feed or for processing, c. intentional introduction into the environment | The Biosafety Act, 2012 | 31-12-9999 | Good | |
| Labelling Requirement for Genetically Modified Organism | Permit Requirement | Labelling of GMOs 1. A GMO or product of a GMO shall be clearly identified and labelled as such, and the identification shall specify the relevant traits and characteristics given in sufficient detail for purposes of traceability. 2. A GMO or any product of a GMO shall be clearly labelled and shall comply with such requirements as may be imposed by the Competent Authority, to indicate that it is or has been derived from a GMO, and where applicable, whether it may cause allergic reactions or pose other risks. | Labeling requirements are supported by documentation requirements of GMO | The Biosafety Act, 2012 | 31-12-9999 | Good | |
| Genetical Modification Organisms Authorization Requirements | Permit Requirement | A person proposing to export a GMO covered by this Act from the country to another country party to the Cartagena Protocol shall- (a) notify the Competent Authority of the proposed country party of import, in writing, prior to the first transboundary movement of the GMO by supplying, at a minimum, information specified in the first schedule (b) include a declaration that all information provided in such notification is factually correct; (c) prior to shipment, provide to the Competent Authority with a copy of the authorisation granted by the importing country | Authorisation is required under the Cartagena Protocol and the applicable laws of the domestic country. | The Biosafety Act, 2012 | 31-12-9999 | Good |